The Monoclonal, Massive Globulin- Waldenstrom Macroglobulinaemia- Juniper Publishers
Juniper Publishers- Journal of cell Science
Preface
Wald Enstrom macrogobulinaemia is a disorder
designated with a nomenclature of a Swedish physician Jan Gosta
Waldenstrom (1906-1996). The exceptional disease was initially scripted
in 1944 [1,2]. Waldenstrom macroglobulinaemia may be defined as the
appearance of a serum para-protein such as immunoglobulin M (Ig M) in
addition to a malignant lymphoplasmacytic infiltrate confined to the
bone marrow. Lymphoplasmacytic Lymphoma (LPL) may be cogitated as a
neoplasm comprising of miniature B lymphocytes, plasmacytoid lymphocytes
and mature plasma cells. The tumefaction generally implicates the bone
marrow with an occasional presence in the lymph node and spleen.
Lymphoplasmacytic lymphoma is accompanied by Waldenstrom
macroglobulinaemia in a majority (95%) of instances [1,2]. The dual
conditions may be denominated by an immunoglobulin M (Ig M) monoclonal
gammopathy accompanied by an emergence of a lymphoplasmacytoid lymphoma
restricted to the bone marrow. Lymphoplasmacytoid lymphoma may concur
with an infection of hepatitis C virus (HCV). A familial prevalence may
be delineated. An estimated 1.4% of neoplasm of miniature B lymphocytes
may be cogitated by lymphoplasmacytoid lymphoma [1,2].
Disease Characteristics
Lymphoplasmacytic lymphoma may be categorized as a
post germinal centre B cell (CD10-, MUM 1+ and BCL6+/-) lymphoma
commingled with divergent plasmacytic cellular differentiation. The
lymphoma may depict concomitant infection with Hepatitis C Virus (HCV).
The bone marrow infiltrate may predominantly be interstitial, nodular or
of a diffuse configuration. Bone marrow trephine biopsy and bone marrow
aspirate may demonstrate an admixture of miniature lymphocytes,
plasmacytoid lymphocytes and mature plasma cells [2,3]. The malignant
cellular egress may enunciate a monotypic secretion of serum
immunoglobulin M (Ig M) protein which may be elucidated in a majority
(> 90%) of instances. Subjects with Waldenstrom macroglobulinaemia
frequently depict vascular hyper-viscosity. Thus, Waldenstrom
macroglobulinaemia may be cogitated as a non-Hodgkin’s lymphoma
concomitant with lymphoplasmacytic lymphoma asmajority (95%) of subjects
of lymphoplasmacytic lymphoma elucidate features of Waldenstrom
macroglobulinaemia. The indolent lymphoplasmacytic lymphoma and
concomitant Waldenstrom macroglobulinaemia may exemplify a disorder of
obscure origin [2,3]. Associated aspects of probable disease insurgence
may be
1. Male sex
2. Enhancing age of disease emergence with a median age of diagnosis at 65 years,
3. A racial predisposition in Caucasians
4. The concurrence of immunoglobulin M monoclonal gammopathy of undetermined significance (Ig M MGUS).
Waldenstrom macroglobulinaemia may progress to
adjunctive B lymphocyte malignancies with an estimated proportion of 10%
at 5 years, 18% at 10 years and 24% at 15 years of disease incurrence
with an overall ratio of malignant conversion at 1.5 % per year. The
neoplasm also displays a familial preponderance and nearly 20%
individual’s manifest family members suffering from Waldenstrom
macroglobulinaemia and associated B lymphocyte malignancies.
Environmental factors such as exposure to radiation or agent orange,
hazardous occupation with handling leather, rubber, paints, dyes and
solvents, coexistent autoimmune disease and infection with hepatitis C
virus (HCV) may be incriminated in the evolution of the malignancy
[3,4].
Clinical Elucidation
The circulation of serum monoclonal immunoglobulin M
(Ig M) in Waldenstrom macroglobulinaemia may display characteristic
constitutional symptoms with concurrent deposition of monoclonal
immunoglobulin M (Ig M) protein in several body tissues with a
consequent emergence of auto-antibodies. Waldenstrom macroglobulinaemia
may manifest systemic symptoms with an estimated serum monoclonal
immunoglobulin M (Ig M) protein greater than 3 grams/decilitre and a
bone marrow ingress of malignant lymphoplasmacytoid
cells greater than 20%. Approximately one fourth
(27%) instances of Waldenstrom macroglobulinaemia may
be asymptomatic, with anaemia in roughly 38% subjects, the
emergence of hyper-viscosity in around 31% individuals, the
appearance of B symptoms (fever, weight loss, night sweats) in
nearly 23% and neurological symptoms in about 22% of patients
[1,2]. Waldenstrom macroglobulinaemia may depict specific
complications such as hyper-viscosity, tissue aggregation
of immunoglobulin M (Ig M) or autoimmune haemolysis
secondary to circulating macro-globulins. Subjects may present
with haematemesis, haemorrhage from the nasal cavity and
retinal vasculature [5,6]. Anaemia, thrombocytopenia, elevated
Erythrocyte Sedimentation Rate (ESR), lymph node enlargement
and hepato-splenomegaly may ensue. A bone marrow trephine
biopsy may exemplify an abundance of malignant lymphoid
cells. Radiographic analysis of the implicated bones may be
unremarkable, thereby excluding a multiple myeloma. Serum
protein examination may detect the presence of an extremely
high molecular weight protein,” a macroglobulin”, cogitated
as an excess of immunoglobulin M [5,6].The quantification
of monoclonal immunoglobulin M (Ig M) may be concordant
with the magnitude of bone marrow infiltration and severity
of systemic symptoms. Hyper- viscosity may appear as chronic
haemorrhage from the nasal cavity, gingiva or gastrointestinal
tract accompanied by headache, dizziness, loss of coordination
or balance, impaired hearing with tinnitus with blurring or loss
of vision. Retinopathy may ensue on account of distended retinal
veins and swelling of the optic disc. Severely affected subjects
may display manifestations of heart failure, drowsiness, stupor
and coma. Systemic symptoms may be discerned at a quantifiable
serum Ig M value greater than 4000 milligrams/decilitre, though
immunoglobulin levels may vary. Constitutional or B symptoms
such as fever, weight loss greater than 10% of the body weight in
preceding six months, drenching night sweats and fatigue may
appear [6,7]. Peripheral neuropathy may be a manifestation of
the disorder. Cold agglutinin disease may occur on account of
elevated circulating antibodies to red blood cells which may
aggregate at minimal body temperatures and induce a haemolytic
anaemia along with Raynaud’s phenomenon, jaundice and
haemoglobinuria. Cryoglobulinemia may be encountered with
the precipitation of immunoglobulin M (Ig M) at reduced body
temperatures in order to obstruct the miniature blood vessels
with emerging consequences such as Raynaud’s phenomenon,
thrombocytopenic purpura, haemorrhaging ulcers and gangrene
of the fingers, toes, nose and ears [1,2].
Amyloidosis may occur with the configuration of an
anomalous “amyloid” protein” which may accumulate in tissues
and organs of the body such as gastro-intestinal tract, renal and
hepatic tissue or heart and peripheral nerves. Malfunctions such
as carpal tunnel syndrome, malabsorption, macroglossia, dermal
thickening, swelling of the extremities, congestive heart failure
and renal failure may emerge. “BING NEEL” syndrome may be
cogitated with a lymphoplasmacytic infiltrate or deposition of
immunoglobulin M (Ig M) within the central nervous system
(brain or spinal cord). Systemic symptoms such as mental
deterioration, confusion, visual disturbance, irritability, altered
personality, convulsions and coma may concur. Recurrent
sinus and upper respiratory tract infection, pleural effusion,
pulmonary infiltrates and occasional rash may be delineated.
Tumour cells of lymphoplasmacytic lymphoma may configure
nodular aggregates in the skin, extremities, spine, breast and
orbital socket [6,7].
Morphological Elucidation
Waldenstrom macroglobulinaemia with coexistent
lymphoplasmacytic lymphoma enunciates malignant cells
with characteristics of B lymphocytes and plasma cells,
denominated as lymphoplasmacytic cells. A diffuse or interfollicular
proliferation of malignant lymphoid cells may
be cogitated. Cellular aggregates devoid of proliferation
centres may be proportionately constituted by miniature B
lymphocytes, plasmacytoid lymphocytes and mature plasma
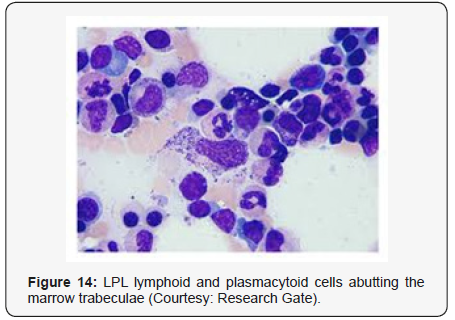
cells. A predominant lympho-plasmacytic infiltrate may be
situated in the inter-trabecular region of the bone marrow [3].
Peripheral blood picture concordant with acute leukaemia may
be demonstrated in an estimated one third (30%) instances.
Tumour cells may predominantly omprise solely of miniature
lymphocytes or small, mature lymphocytes commingled with
plasmacytoid lymphocytes. The bone marrow may be infiltrated
by an identical malignant infiltrate. Mature plasma cells, tissue
mast cells and histiocytes may be quantifiably augmented.
Plasma cells may infrequently be the preponderant cellular
component. Serial sections from bone marrow trephine biopsy
may delineate a diffuse or a focal lesion.The focal lesions may
configure a para trabecular, interstitial or non paratrabecular
pattern of tumour incrimination [3]. Expansive marrow
replacement by the tumefaction may induce a significant
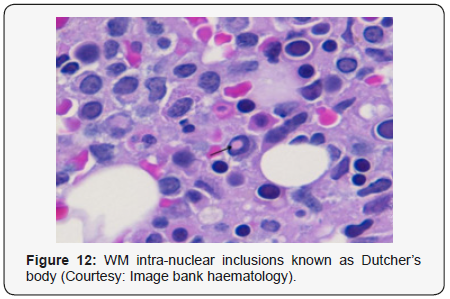
reduction of normal haematopoiesis. Intra-nuclear inclusions
termed as “Dutcher’ s bodies” may be articulated within
lymphocytes and plasma cells and may be considered diagnostic
of lymphoplasmacytic lymphoma. Intra-nuclear inclusions
reactive to Periodic Acid Schiff’s (PAS) stain may similarly be
configured in plasma cells constituting a multiple myeloma or
reactive lymphoid proliferations. Lymphoplasmacytic lymphoma
with Waldenstrom macroglobulinaemia may terminate with the
evolution of a Richter’s syndrome, thereby recapitulating a Small
Lymphocytic Lymphoma (SLL). The malignant egress may lack
the presence of monocytoid cells, in contrast to a marginal zone
lymphoma. The occurrence of Dutcher ‘s bodies with admixed
enlarged, transformed lymphocytes may be characteristic of
lymphoplasmacytic lymphoma. Mast cells may be intermingled
with epitheloid histiocytes. Intercellular material stained with
periodic acid Schiff (PAS+) stain may be exemplified along with
scattered amyloid and crystal engulfing histiocytes [7,8].
Immune phenotype
Lymphoplasmacytic lymphoma cells may exemplify a
surface immunoglobulin M (Ig M+). Immunoglobulin molecules
confined to the cytoplasm may primarily be immunoglobulin M
(Ig M) although Ig G or infrequently Ig A may be elucidated. The
lymphoma may be immune reactive to CD20+ and associated
pan B lymphocyte antigens such as CD19+, CD79a+ and PAX5+.
However, a percentage may be non eactive for the aforementioned
immune markers. Nonreactive immune molecules may be CD5-,
CD10-, CD23- and BCL6- although CD5 may be debatable (-/+)
(1,3). Plasmacytic immune markers CD38 and CD138 may be
equivocal (+/-) in specific instances.
Molecular Characterization
A frequent genomic abnormality the MYD88L265P mutation
may be discerned in a majority (95%) along with chromosomal
deletion of del [6] (q21). Chromosomal translocation t (9:14)
may be exceptional. The MYD88L265P chromosomal mutation
may be universal in Waldenstrom macroglobulinaemia. A whole
genome sequencing may depict the mutation in 90% instances.
MYD88L265P chromosomal mutation may be infrequent in
multiple myeloma, marginal zone lymphoma or immunoglobulin
M –monoclonal gammopathy of undetermined significance (Ig
M MGUS). Chromosomal mutation CXCR4 may be enunciated
which may be identical to the WHIM syndrome (warts,
hypogammaglobulinaemia, infections and myelokathexis).
Individuals with Waldenstrom macroglobulinaemia devoid of
MYD88 or a CXCR4 chromosomal mutation may depict an inferior
survival, in contrast to instances delineating the mutations [1,2].
Differential Diagnosis
Lymphoplasmacytic lymphoma may necessitate a distinction
from Chronic Lymphocytic Leukaemia (CLL), mantle cell
lymphoma and plasmacytoid variants of extra nodal or nodal
marginal zone lymphoma. Chronic Lymphocytic Leukaemia
(CLL) may exhibit a focal plasmacytic differentiation. The tumour
cells may be immune reactive to CD5+, CD23+ and a CD20 dim, in
contrast to a lymphoplasmacytic lymphoma. Immunoglobulin M
(Ig M) para-protein may be absent or minimal. Splenic marginal
zone lymphoma may demonstrate an intra-sinusoidal pattern
of marrow incrimination. Plasmacytic differentiation may be
reduced or minimal. Immunoglobulin M (Ig M) para-protein may
be lacking or be of miniscule quantities [1,2]. Distinction from
plasmacytoid lymphoma may be particularly cogitated with
demonstration of plasma cells and plasmacytoid lymphocytes,
manifesting numerous inclusions confined to the cytoplasm
which may react to the Periodic Acid Schiff ‘S (PAS+) stain. The
tumour cells may thus recapitulate the appearance of histiocytes.
Chromosomal point mutation MYD88 may be elucidated in a
majority (90%) of instances of lymphoplasmacytic lymphoma,
contrary to an exceptional delineation in multiple myeloma and
marginal zone lymphoma [8,9] (Figures 1-14).
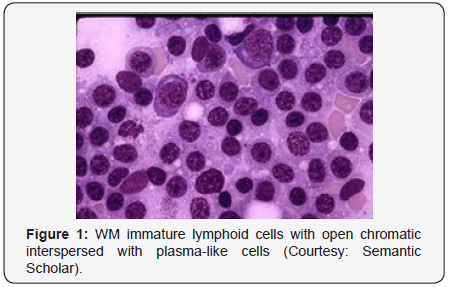
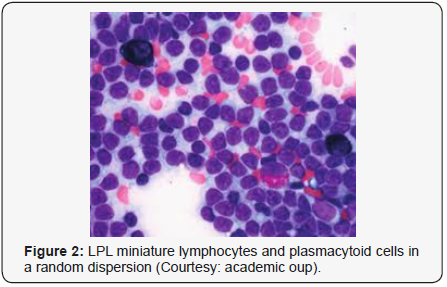
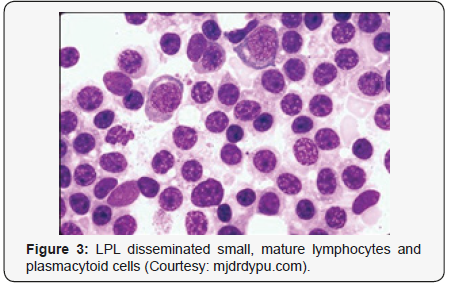
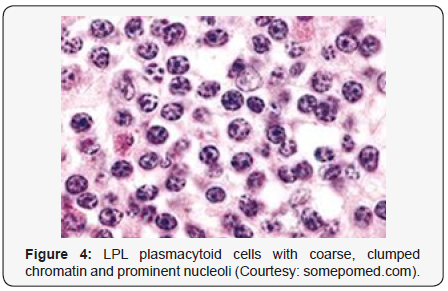
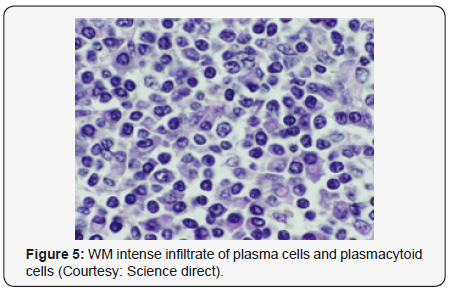

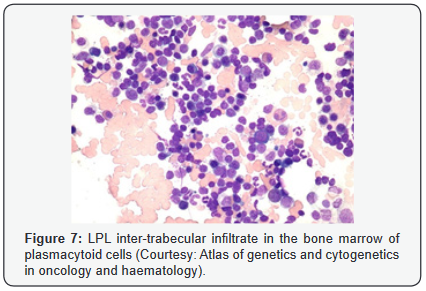

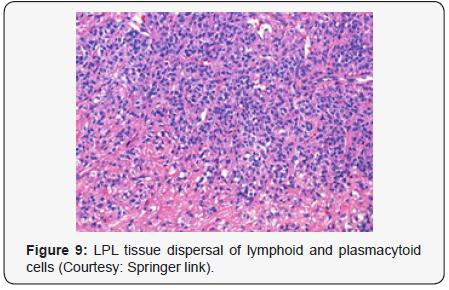
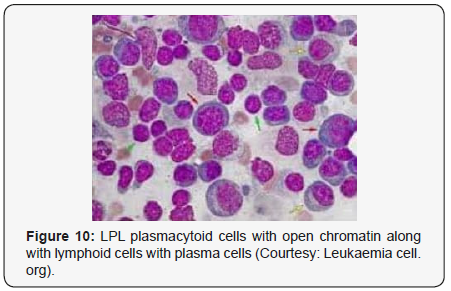


Criterion for Discerning Variants of Waldenstrom Macroglobulinaemia
1. A monoclonal gammopathy with Immune Globulin
M (IgM) irrespective of the magnitude of M protein and
an infiltration of lymphoplasmacytic cells greater than
10% in the bone marrow may be elucidated. Particularly
inter-trabecular tumour dissemination may comprise of
miniature lymphocytes with a plasmacytoid or plasma cell
differentiation and a characteristic immune phenotype
of immune reactive surface immune globulin M (Ig M+),
CD19+, CD20+ and nonreactivity for CD5-, CD10-and CD23-
. The aforementioned evaluation may competently exclude
adjunctive lympho proliferative disorders such as Chronic
Lymphocytic Leukaemia (CLL) and mantle cell lymphoma
[1,2].
2. A monoclonal gammopathy of undetermined
significance (Ig M MGUS) may enunciate serum monoclonal
immunoglobulin M (Ig M) protein values below 3 grams/
decilitre with a lymphoplasmacytic cellular infiltrate
beneath 10% generally confined to the bone marrow.
Systemic symptoms of anaemia, hyper-viscosity, lymph
node enlargement or hepato-splenomegaly may be
absent. Immunoglobulin M monoclonal gammopathy of
undetermined significance (Ig M MGUS) may evolve into a
florid Waldenstrom macroglobulinaemia or an adjunctive
B lymphocyte malignancy. The proportion of malignant
transformation may emerge at an estimated 1.5% every year.
3. Smouldering Waldenstrom macroglobulinaemia
may be an indolent or asymptomatic disorder. Monoclonal
serum protein values for immunoglobulin M (Ig M)
exceeding 3 grams/decilitre and/or a lymphoplasmacytic
infiltrate restricted to the bone marrow in excess of 10%
may be enunciated. End organ damage with coexistent
anaemia, hyper-viscosity, lymph node enlargement or
hepato-splenomegaly on account of a lymphoplasmacytic
proliferation may be absent [1,2].
Investigative Assay
Discernment of Waldenstrom macroglobulinaemia
mandates a disease evaluation with complete blood count,
serum chemistries such as liver and renal function tests, blood
glucose and specific procedures such as bone marrow trephine
biopsy and bone marrow aspiration. Serum immunoglobulin
assay may depict an overproduction of immunoglobulin M with
a decline in the values of immunoglobulin G and A (Ig G and I
gA), feature which may enhance the probability of emergent
infections. Radiographic investigations may include a plain X-ray,
Computerized Tomography (CT) scan, a Magnetic Resonance
Imaging (MRI), an ultrasound and Positron Emission Tomography
(PET) scan of the lymph node enlargement, enlarged spleen and
dermal or tissue infiltrates of lymphoplasmacytic lymphoma
cells [9,10]. Ocular examination may incorporate the assessment
of retina and ocular fundus.
Therapeutic Options
Commencement of therapeutic intervention may concur
with the appearance of B symptoms such as fever, night sweats,
weight loss, fatigue, lymph node enlargement or splenomegaly.
Haemoglobin declining to below 10 grams/decilitre or a
platelet count below 100,000/ microlitre may be cogitated on
account of bone marrow infiltration. Additionally, complications
such as hyper viscosity syndrome, symptomatic sensory or
motor peripheral neuropathy, systemic amyloidosis, renal
insufficiency or symptomatic cryoglobulinaemia may require
therapy [10,11]. A policy of careful observation may suffice for
the indolent disorder. An estimated half (50%) of the subjects
with Waldenstrom macroglobulinaemia devoid of constitutional
symptoms and a lack of treatment at 3 years following diagnosis
may be managed with “active surveillance “. Approximately
10% instances may not require a therapeutic intervention
during a 10year duration following detection. It may be crucial
to establish the emergence of a hyper-viscosity syndrome
prior to initiation of therapy and if plasmapheresis may be
necessitated. Waldenstrom macroglobulinemia when associated
with hyper-viscosity may display systemic symptoms such as
visual deterioration, neurological symptoms and haemorrhage,
incurring with immunoglobulin M serum values greater than
4 grams/decilitre and the condition may be benefitted with
plasmapheresis [11,12]. Chemotherapeutic agents found
efficacious in Waldenstrom macroglobulinaemia include
chlorambucil, cyclophosphamide, bendamustine, nucleoside
analogues such as fludarabine and cladribine. Corticosteroids
prednisone or dexamethasone may be applicable. Biologic
therapy may enunciate the utilization of anti monoclonal
antibody conjugates such as rituximab, ofatumumab or
obinutuzumab. Immune modulators such as thalidomide and
lenalidomide may prove to be effective [1,2]. Administration
of proteasome inhibitors such as bortezomib, carfilzomib and
ixazomib may be advantageous. Targeted therapy implicating
the B cell signalling pathway may concur as imbruvica and
everolimus [12,13]. Initial or preliminary therapy for previously
untreated, symptomatic subjects may employ
a. purine analogues with a combination of fludarabine,
cyclophosphamide and rituximab (FCR) or fludarabine and
rituximab (FR).
b. Alkylating agents in varying combinations such as
rituximab with cyclophosphamide, doxorubicin, vincristine
and prednisone (R CHOP), dexamethasone, rituximab and
cyclophosphamide (DRC) or rituximab with bendamustine
(BR) may be beneficial
c. Bortezomib in diverse combinations such as
bortezomib, dexamethasone and rituximab (BDR) may be
applicable.
d. Singular antibody conjugate such as rituximab may be
utilized for initiation of therapy.
e. Ibrutinib as a Bruton’s tyrosine kinase inhibitor (BTK
inhibitor) may be efficacious in instances with MYD88
mutation. Concomitant chemo immunotherapy may be
administered
Administration of bendamustine with rituximab may
depict a median and inter-quartile range (IQR) of 69.5 months
in patients of Waldenstrom macroglobulinaemia [1,2]. The
application of R CHOP may display a median and inter-quartile
range (IQR) of 28.1 months. Solitary administration of rituximab
in Waldenstrom macroglobulinemia may exhibit an objective
response rate (ORR) of 52%, a partial response (PR) of 27% and
a minor response (MR) of 25%. The median duration of response
(DOR) may be elucidated at 27 months. An estimated half (54%)
of the individuals may depict an elevated immunoglobulin M
(Ig M) “flare” and one fourth (27%) subjects may delineate a
persistent elevation of serum immunoglobulin M at 4 months
duration following discernment of disease. Administration of
ibrutinib may demonstrate an objective response rate (ORR)
of 90.5%, a partial response (PR) of 73% and the median time
to suitable therapeutic response may appear at 4 weeks. The
progression free survival (PFS) at the end of 2 years may be
69.1% and Overall Survival (OS) at 95.2 %. Toxicity levels to the
chemotherapeutic agent may be greater than grade 2. Ibrutinib
administration may be accompanied by specific toxicities such
as thrombocytopenia, neutropenia, stomatitis, atrial fibrillation,
diarrhoea, herpes zoster infection, haematoma, secondary
hypertension and epistaxis [13].
Contemporary Instances
Contemporary instances of Waldenstrom
macroglobulinaemia may necessitate management as described:
1. Monoclonal gammopathy of undetermined significance
(Ig M MGUS) with a lymphoplasmacytic infiltrate below
10%, an asymptomatic or smouldering Waldenstrom
macroglobulinaemia with haemoglobin greater than 11
grams/decilitre, a platelet count in excess of 120,000/
millilitre may be managed by” watchful waiting”.
2. A symptomatic Waldenstrom macroglobulinaemia with
a haemoglobin level below 11 grams/decilitre or a platelet
count beneath 120,000/ millilitre.
a. Immunoglobulin M (Ig M) related neuropathy.
b. Haemolytic anaemia secondary to
Waldenstrom macroglobulinaemia.
c. Symptomatic cryoglobulinaemia. The
aforementioned instances may be managed with a solitary
antibody conjugate such as rituximab. A maintenance
therapy may not be required. Plasmapheresis may be
opted for in instances of hyper-viscosity secondary to
chemotherapy [1,2].
3. Waldenstrom macroglobulinaemia elucidating bulky
disease (tumour magnitude greater than 10 centimetres)
or profound pancytopenia with reduced blood counts such
as haemoglobin below 10 grams/decilitre or a platelet
count beneath 100,000/ millilitre with the appearance
of constitutional symptoms and a lack of features of
hyper-viscosity or hyper-viscosity may be managed with
plasmapheresis.
The aforementioned subjects may be administered
concomitant bendamustine with rituximab with the regimen
devoid of maintenance therapy with singular rituximab. Stem
cell transplantation may be a pre-requisite. Alternatively, stem
cells may be mobilized and cryo-preserved for subjects beneath
≤ 60 years of age or emerge as potential and future candidates
of Autologous Stem Cell Transplantation (ASCT). Stem cell
transplantation may be suitably employed with subjects of
relapsed or refractory Waldenstrom macroglobulinaemia.
Autologous stem cell transplant (ASCT) may depict a 5year
Progression Free Survival (PFS) of 40% and an overall survival
(OS) of 68%. Allogeneic stem cell transplant may exhibit a 5year
progression free survival (PFS) of 56% and a 5year overall
survival (OS) of 62% [1,2]. Clinical trials with novel agents or
drug conjugates may be mandated. Contemporary drugs such
as ibrutinib, a Bruton’s tyrosine kinase inhibitor or idelalisib, a
PI3kinase inhibitor or everolimus, an m TOR inhibitor may be
efficaciously adminstered. Contemporary anti CD20 antibody
conjugates such as of tamumab, anti BCL2 agents such as
venetoclax, recent histone deacetylase (HDAC) inhibitors such
as panobinostat, recent proteasome inhibitor carfilzomib,
recent immunomodulatory agents such as pomalidomide may
be additionally assessed. Contemporary targeted therapies may
include molecules such as ventoclax, acalabrutinib and BGB3111.
The aforementioned drugs may be utilized in combination with
established agents.
Salvage Therapy
Salvage Therapy may be applicable in specific instances.
In subjects where the requirement of subsequent therapy
may exceed 4 years, the original therapeutic regimen may be
replicated. For therapeutic installation within 4 years, a monotherapy
with ibrutinib or combinations such as Dexamathasone,
Rituximab and Cyclophosphamide (DRC) may be opted for.
Concomitant administration of bendamustine with rituximab
(BR), Bortezomib, Dexamethasone and Rituximab (BDR) may be
effective.
Supportive Therapy
Supportive Therapy may incorporate modalities such as
blood transfusion, administration of growth factors in order to
enhance the blood cell counts (white and red blood cells with
platelets). Surgical procedures may be specified in particular
instances in the form of splenectomy or plasmapheresis in
order to reduce the serum immunoglobulin M (Ig M) quantities.
Targeted radiation may be employed in order to decimate the
magnitude of incriminated lymph nodes [12,13] Table 1.
Table 1: Distinction betwixt Lymphoplasmacytic Lymphoma (LPL) and small cell Plasma Cell Myeloma (PCM) [1,2].
For more Open access journals
please visit our site: Juniper Publishers
For more articles please click
on Journal of Cell Science & Molecular
Biology




Comments
Post a Comment